Stable Isotope Analysis - the basics
Elements and isotopes
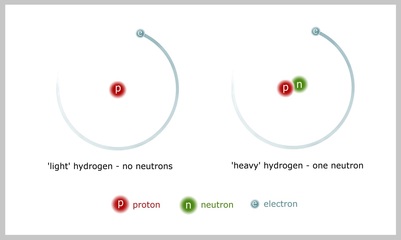
Elements are the simple chemical building blocks of everything around us. Nitrogen, oxygen and gold are just a few examples. Each element consists of three components – the protons and neutrons that form the nucleus and the electrons that move around it. The number of protons is what makes an element what it is; any element with 8 protons is always going to be oxygen. However, many elements such as hydrogen, oxygen, and carbon, take multiple forms.
|
Different forms of the same element are called isotopes of that element. Isotopes of a particular element react chemically in the same way, but they vary in their number of neutrons. This means that isotopes differ ever so slightly in weight. Hydrogen, for example, has an isotope with one proton and no neutrons (light) and an isotope with one proton and one neutron (heavy). Both these isotopes of hydrogen can be found together in any body of water, such as the sea, or the water we drink.
NOTE: There are two types of isotopes- those that are stable and those that are radioactive. Radioactive isotopes decay over time and are commonly used for dating material, for example in radiocarbon dating, Stable isotopes on the other hand do not decay; as the name implies, they are stable. |
Because they differ in weight, heavy and light isotopes move through food chains, and systems such as the water cycle, at different rates. The ratio of heavy to light isotopes therefore varies between food types and geographic locations. By measuring these ratios (or isotopic signatures) we can find out where an animal has been and what it has eaten.